Bioremediation of Binary System of Reactive Red 120 Dye and Cr(III) Using Aspergillus tamari and Statistical Validation of Response
Copyright © 2019 by Asian Journal of Atmospheric Environment
This is an open-access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
The efficiency of Aspergillus tamarii (isolated from sludge of a textile industry) was investigated to remediate the synthetic solutions of reactive red 120 (RR120) dye and chromium(III) separately. Also, parallel studies were conducted on the bioremediation of binary system of chromium(III) and RR120 dye in different ratios. The study was conducted to find the potential of the A. tamarii for removing chromium(III) and color from solutions containing only chromium or only RR120 dye or both and the effect of RR120 and chromium on the growth of the strain were observed. Maximum dye removal 81 mg/L was observed from 100 mg/L RR120 dye solution at pH 5 up to 50 hours. Maximum chromium removal 88.3 mg/L was observed from 100 mg/L chromium (III) solution at pH 5 up to 50 hours. In previous studies, A. tamarii was used as a bioremediator for removing different chromium complex dyes. The removal of chromium(III) and color were compared from solutions of synthetic mixtures and chromium complex dyes. The response of bioremediation study was validated using multiple regression analysis.
Keywords:
Binary system, A. tamarii, Chromium(III), RR120 dye, Statistical Validation1. INTRODUCTION
Chromium is one of the most frequently found heavy metal pollutants (contaminants) in groundwater at hazardous waste sites. Chromium is found in the nature in either of the two forms - Cr(VI) and Cr(III). The compounds of chromium are irritants and easily absorbed by the lungs. Also, intense tracheo-bronchial irritation were observed after the inhalation of chromium-compounds (Baruthio, 1992; Sullivan, 1969). The Cr(VI) form is generally present in the form of chromate (CrO4-) and dichromate (CR2O7-) and shows notably higher levels of toxicity than other valence states (Horsfall et al., 2006). There are a large number of processes like leather tanning, steel manufacturing, electroplating, corrosive paints, photographic material, wood preservation and so on that releases chromium into the environment (Miretzky and Cirelli, 2010). The leaching from rocks and topsoil is the most common natural source of chromium contamination into waterbodies (Mills et al., 2011; Bertolo et al., 2011). Further, the concentrations of chromium in the soil may increase mainly via atmospheric depositions and improper disposal of liquid and solid waste from chromate-processing industries. Due to its persistent nature, chromium bio-accumulates in the food chain by long time exposure, thus causing toxic health hazards like irritation in the lungs as well as the stomach, cancer in the digestive tract, less growth of plants and also death in animals (Sen et al., 2011, 2005; Sari et al., 2008). Also it is worth mentioning that, the contamination of surface water by synthetic colours is viewed to be another environmental issue (Wu et al., 2012). Synthetic dyes with various structural forms like acidic, azo, basic, diazo, disperse, reactive, metal-complex based and anthraquinone-based are commonly utilized as colouring agents in cosmetic, leather, paper, food and textile industries. Further, chromium complex dyes are commonly used in woollen and nylon industries (Ghosh et al., 2017). The effluents from these industries are highly toxic due to the presence of dye and heavy metal. Additionally, the disposal of these effluents into the receiving water bodies has hazardous impacts on the flora, fauna and other forms of life dependent on these water bodies.
The common physico-chemical treatment techniques used in large scale industries for the removal of metals and dyes from the wastewaters include ion-exchange, membrane processes, adsorption, etc. which have some economic and technical constraints. The removal of heavy metals and dyes using microorganisms (algae, bacteria and fungi) has gained prime importance as a possible option to the already existing physico-chemical methods for the treatment of heavy metal/dye contaminated effluents (Bachate et al., 2013; Puyen et al., 2012). Also the bioremediation process is environmental friendly, requires less energy and produces less chemical sludge than the conventional processes (Saroj et al., 2014). Existing studies have demonstrated that Fungi shows higher tolerance towards dyes/metals than other micro-organisms and has higher capacity to remove pollutant due to higher surface to volume ratio (Mishra and Malik, 2014; Skorik et al. 2010; Sun and Shao 2007; Fomina et al., 2007; Guimaraes-Soares et al., 2006; Akar and Tunali 2005). It is also worth mentioning that fungi appears to be most suitable in the treatment of effluents contaminated with dyes and metals due to its capability to excrete different oxidoreductive enzymes (Ghosh et al., 2015).
A. tamarii isolated from the sludge of a textile industry in the laboratory of the present authors was studied (previously reported) to reduce different trivalent chromium complex dyes (commonly used in leather and textile industries) namely acid blue 158, acid brown 45, acid black 52, acid orange 80 and acid orange 86 dyes from synthetic solutions and actual industrial effluents (Ghosh et al., 2017, 2016, 2014). From previous studies, it was also observed that acid blue 158 dye has higher chromium(III) contents than the above mentioned chromium complex dyes (Ghosh et al., 2017, 2016, 2014). In the present study, we compare our findings of single and and binary systems with the findings of acid blue 158 dye. The removal of chromium and dye and the growth of A. tamarii were reported to be dependent on the combined effect of both chromium and dye. However, relatively fewer studies are available on the removal of chromium alone or dye alone using the same organism. Further, actual industrial effluents may contain both chromium and dye in different combinations, whereas the competitive interaction of both chromium and dye in solution affecting the growth of organism and the removal of chromium and dye is very little understood. In the present study, the potential of A. tamarii to grow and remove chromium(III) and dye (RR 120) from solutions containing only chromium(III), only dye and binary system of both in different proportions in each system was examined in batch bioreactors using synthetic solutions of chromium and dye. The binary system better represents the closer composition of industrial effluents and the studies with the same shall provide better understanding of the competitive interaction of chromium and dye and their effects on the growth of fungus and the removal of chromium and dye.
2. EXPERIMENTAL
2. 1 Preparation of Dye and Chromium(III) Solutions
Reactive red 120 (RR120) dye (commercial name: Jokazol red) is commonly used azo dye in the textile industry and was chosen for the present study. The chemical structure of the dye and CAS number (61951-82-4) are reported online (Website: https://pubchem. ncbi.nlm.nih.gov/compound/Reactive-Red-120). The synthetic stock solution (100 mg/L) of dye was formulated in the laboratory by dissolving required quantity of powdered reactive red 120 (RR120) dye in distilled water. The water-soluble dye was used without any purification. Similarly, the synthetic stock solution (100 mg/L) of chromium(III)was prepared by dissolving appropriate quantity of chromium nitrate reagent. The solutions of various concentrations of dye, chromium( III) and the binary system were made by appropriate dilution of the stock solutions. All the chemicals and reagents (AR grade) were obtained from E. Merck (India) Ltd., HiMedia Laboratories Pvt. Limited and Qualigens Fine Chemicals.
2. 2 Microorganism and Growth Media
The Fungi A. tamarii isolated from the sludge of a textile industry appeared to be filamentous under microscopic examination and fast growing. It was studied to remove various chromium(III) complex dyes such as acid brown 45, acid black 52, acid orange 80, acid orange 86, and acid blue 158 (Ghosh et al., 2017, 2016, 2014). The growth medium was as follows: K2HPO4: 0.5 g/L; NaCl: 1 g/L; MgSO4: 0.1 g/L; NH4NO3: 0.5 g/L; glucose: 10.00 g/L and yeast extract: 5.0 g/L (Ranjusha et al., 2010). The prepared media was autoclaved and inoculated with A. tamarii. The pH of the media for maximum growth of A. tamarii in the absence of dye or chromium was reported to be 5.0 by Ghosh et al. (2014). Hence, the pH of the media was kept at 5.0 in all batch experiments. Synthetic media containing only chromium(III) and only RR120dye of different concentrations were made by dissolving the stock solutions of each in growth media. Synthetic growth media containing binary system of chromium(III) and RR120dye was prepared in the different combinations (Table 1). From previous studies, it was observed that acid blue 158 dye has higher chromium(III) contents than acid brown 45, acid black 52, acid orange 80 and acid orange 86 dyes (Ghosh et al., 2017; 2016; 2014). In the current study, the similar concentration of chromium( III) and RR120dye was prepared as present in acid blue 158 dye (Table 1). The structural details of Acid Blue 158 dye are available online (https://pubchem.ncbi.nlm.nih.gov/compound/C.I.-Acid-Blue-158_-disodium-salt).
2. 3 Bioremediation Studies
Bioremediation of binary system was performed using A. tamarii by inoculating actively growing cells of A. tamarii in 250 mL conical flasks (containing 100 mL media) at pH 5 and incubated at 27°C under aerobic and shaking condition (110 rpm) in orbitek shaker up to 50 hours. Small portions were taken from the conical flasks after definite time gaps and centrifuged at 4000 rpm (eltek centrifuge, Model no: TC 4100F). The concentrations of chromium and dye were determined using atomic absorption spectrophotometer (AAS) and UV-visible spectrophotometer, respectively. The biomass collected after centrifugation was dehydrated at 60°C temperature in a hot air oven (Ambassador, India) and estimated gravimetrically to determine the biomass concentration. All the bioremediation experiments including microbial inoculation, incubation, growth, and removal studies were conducted in aseptic condition.
2. 4 Assay Techniques
The concentration of chromium(III) was analysed using AAS (Perkin-Elmer AAnalyst 200) after acid digestion of the samples. The UV-Vis spectral analysis (Systronics UV/VIS Spectrophotometer 117, New Delhi, India) of the RR 120 dye in the wavelength range of 200-900 nm before bioremediation showed maximum absorbance at 508.8 nm. The scanning electron microscopy (SEM) analysis of the dried biomass before and after bioremediation was performed using instruments ZEISS EVO Series Scanning Electron Microscope (Bruker). The samples for SEM analysis were put on a stainless-steel stub and thin coatings of gold and carbon, respectively, were plated under vacuum.
2. 5 Statistical Validation
The response of bioremediation study was validated using multiple linear regression model (run on R & R Studio software), where concentration of contaminants were independent variables and concentration of biomass was dependent variable.
3. RESULTS AND DISCUSSION
3. 1 Bioremediation Studies
Bioremediation experiments were performed to determine the color and chromium removal during the growth period of A. tamarii.
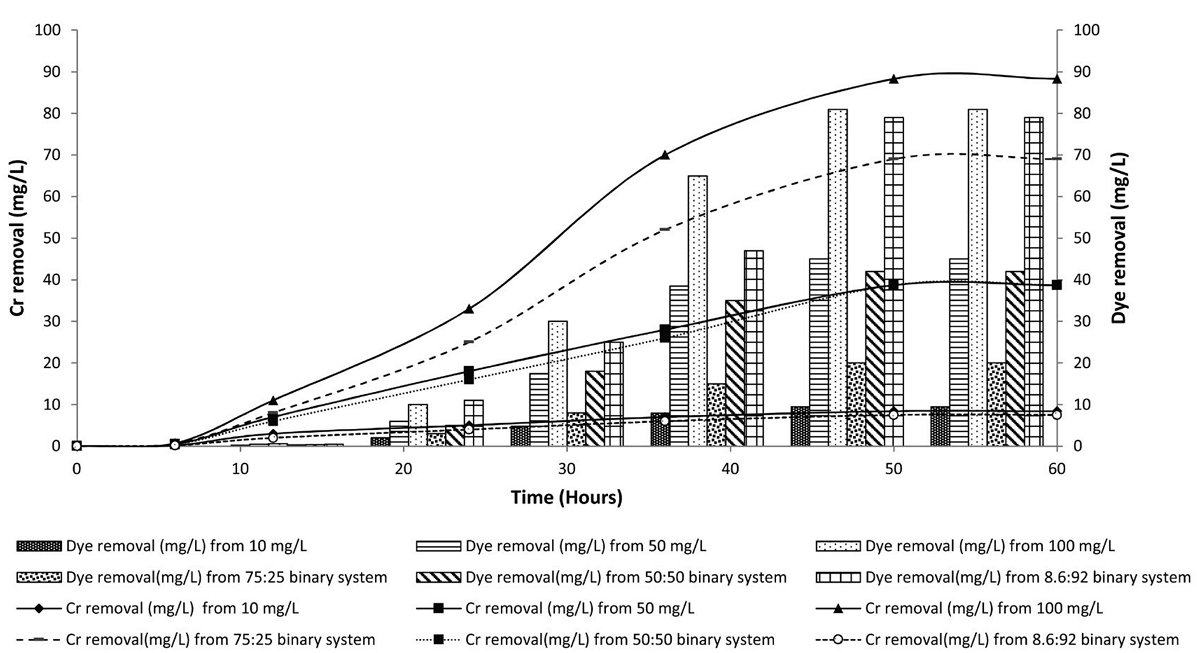
Fig. 1 shows the removal (mg/L) of chromium(III) and dye with time at 10, 50 and 100 mg/L initial concentrations of chromium using A. Tamarii in the batch bioreactor at pH 5 up to 60 hours. The removal of chromium( III) increased up to 50 hours at all initial concentrations of chromium(III), beyond which no improvement was observed. The maximum removal of chromium (88.29 mg/L) was found from 100 mg/L chromium solution. The higher removal of chromium(III) at higher concentration was due to the availability of more chromium. Also during bioremediation studies sets of control (autoclaved) samples were kept in the incubator without the inoculum of A. tamarii. There was no changes observed of the media up to maximum 3 days. This ensures the reactions were due to biological reason.
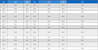
Table 1 shows the removal of dye and chromium from the different synthetic solutions of single and binary systems. Serial nos. 1-3 show single systems of chromium( III) solution at different concentrations. The removal of chromium(III) was noticed to be 88.3 mg/L along with the biomass concentration of 3.4 g/L from 100 mg/L chromium(III) solution. The removal of chromium(III) was found to be 38.7 mg/L along with the biomass concentration of 4.5 g/L from 50 mg/L chromium(III) solution. Similarly, removal of chromium( III) were observed to be 8.4 mg/L along with the biomass concentration of 4.9 g/L from 10 mg/L chromium( III) solution. As the availability of chromium(III) increased in the system, the removal of chromium was increased. The biomass concentrations were observed to be reduced with the increase in concentrations of chromium( III). It is may be due to the toxic effect of chromium( III) at higher concentration on the fungal cells. As shown in Table 1 the biomass concentration was decreased with increase in the concentration of chromium (III) because of the toxic effect of chromium(III) at higher concentration on the fungal cells. The biomass concentration (3.4 g/L) was obtained from 100 mg/L chromium solution.
Similar patterns of the removal (mg/L) of reactive red 120 dye (without chromium complexation) were observed under the same conditions (Fig. 1). The maximum dye removal (81 mg/L) was observed from 100 mg/L dye solution.
Similarly, Table 1, Serial nos. 4-6 show single systems of RR120 dye solution at different concentrations. The removal was observed to be 81 mg/L along with the biomass concentration of 3.2 g/L from 100 mg/L dye solution. The removal of dye was found to be 45 mg/L along with the biomass concentration of 4.1 g/L from 50 mg/L dye solution. The removal of dye was observed to be 9.5 mg/L along with the biomass concentration of 4.8 g/L from 10 mg/L dye solution. Similarly, higher removal of dye was observed in higher concentrations of dye solutions. The biomass concentrations were observed to be reduced with the increase in concentrations of dye.
The Table 1, serial nos. 7-9 show binary systems of chromium(III) and RR120 dye at different concentrations (Fig. 1). The removal of chromium and dye were observed to be 69 mg/L and 20 mg/L from binary system of 75:25 (chromium: dye, mg/L) along with the biomass concentration of 3.6 g/L. The removal of chromium and dye were observed to be 38.73 mg/L and 42 mg/L from binary system of 50:50 (chromium: dye, mg/L) along with the biomass concentration of 3 g/L. The removal of chromium and dye were observed to be 7.5 mg/L and 79 mg/L from binary system of 8.6:92 (chromium: dye, mg/L) along with the biomass concentration of 3.8 g/L. The biomass concentration was reduced with increasing the total concentrations of Cr (III) and dye at binary system. This is due to the toxicity of dye as well as trivalent chromium to the fungal cells at higher concentration.
The lower removal at higher dye concentration may be due to the lower biomass for toxicity effect and the crowding effect of dye molecules on the binding sites of the cells (Zhang et al., 2016; Rakhshaee, 2011). The experimental results (Table 1) also show lower removal at higher concentrations of dye. Further, the lower biomass concentration obtained in the 50:50 binary system and also in 100 mg/L dye solution compared to that obtained in 100 mg/L chromium solution strongly indicates that the reactive red 120 dye has a higher inhibitory effect on biomass concentration than chromium( III). The preferential removal of chromium followed by dye as observed in the present study could be due to the complex structure of reactive red 120 dye.
Table 1, serial nos. 9 and 10 show the removal of dye and chromium along with the biomass concentrations from binary system containing chromium and dye in the ratio of 8.6:92 and 100 mg/L solution of acid blue 158 dye (a chromium complex dye containing chromium 8.6 mg/L), respectively. The higher removal of dye and chromium from the binary system rather than from the solution of chromium complex dye using the same strain at the same initial concentration of the dyes and chromium could be due to the complexity of the structure of the chromium complex dye (serial no. 10). Table 1, serial no. 11 shows the removal of color (86%) and chromium (4.6 mg/L) from the actual textile effluent where chromium complex dyes are used (Ghosh et al., 2017).
Also, higher removal of dye/chromium was observed in the presence of a single pollutant (dye/chromium) rather than both. Lower biomass was noticed in the solution of RR120 dye than chromium(III) solution of same concentration. It may be due to the toxic effect of RR120 dye for complex aromatic structure on the growth of organism. The efficiency of fungus for removing dye/chromium was increased at the lower concentration of dye. Generally, chromium(III) is considered to be less toxic than chromium(VI). Chromium is a heavy metal and cannot be degraded. It may have been adsorbed or precipitated on the cell membrane in the present study (Ghosh et al., 2015).
The higher removal of dye and chromium was observed from synthetic mixture rather than chromium complex dye solution. The lower removal of color and chromium from acid blue 158 dye than binary mixture using the same strain at the same initial concentration of the dyes and chromium could be due to the complexity of the structure of the chromium complex dye.
Table 2 compares the removal potential of A. tamarii (during growth) for color and chromium from synthetic solutions of chromium complex dyes and actual effluent. In previous studies, the lower removal of color and chromium from chromium complex dyes at the same initial concentration of the dye/chromium could be due to the complexity of the structure of the chromium complex dyes. A significant removal of both color and chromium was observed, when dye is not complexed with chromium (Table 1).
In the present study, the SEM analysis of the fungal biomass was performed before and after bioremediation. The SEM micrographs showed cell shape distortion after bioremediation from binary system rather than before bioremediation (Supplementary Figs. 1-3). The SEM study shows the rough cell surface may be due to adsorption or precipitation.
3. 2 Statistical Validation of Results
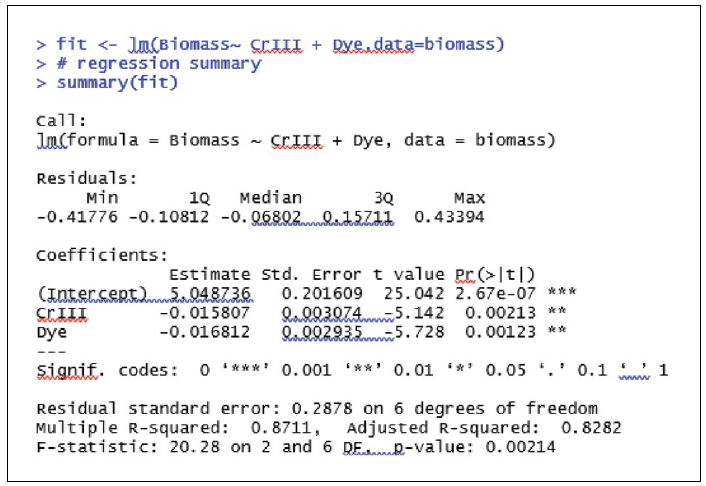
To study the impact of Cr III and Dye on Biomass concentration we run ordinary least square multiple linear regression where “Biomass concentration” is the dependent variable and concentration of Cr III and Dye are the independent variables. R as well as R-studio software have been used to run the multiple linear regression. Data from Table 1 above have been used to run the regression.
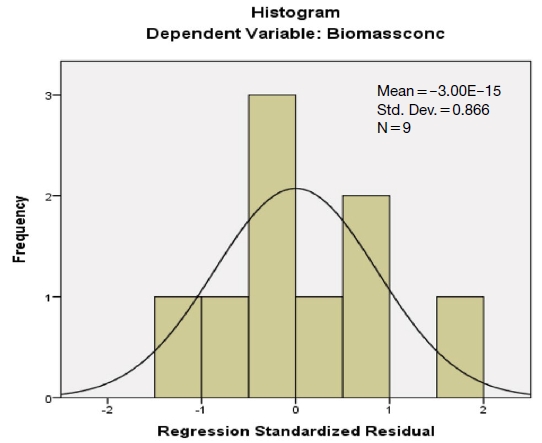
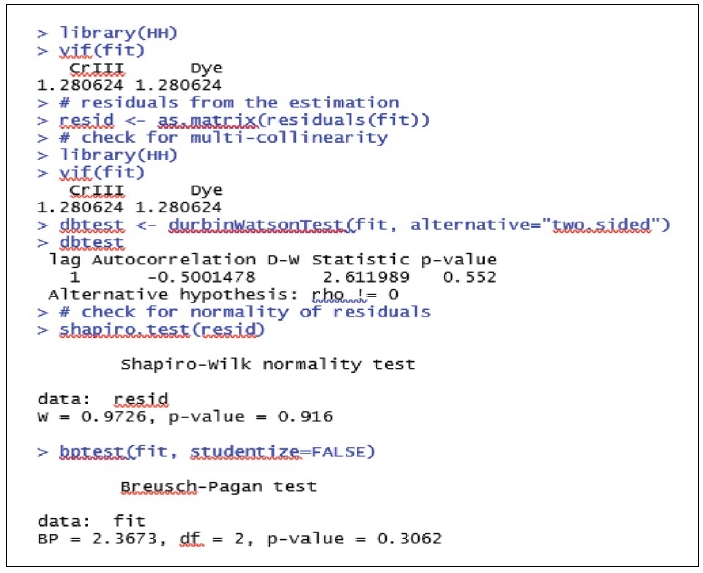
The regression results (Gujarati, 2009) clearly show that the coefficients of the independent variables (concentration of Cr III and Dye) are significant. The significance of the coefficients validates the linear relationship between concentration of Cr III, concentration of Dye and Biomass concentration. The regression model is overall significant (p value associated with F-statistic is less than 0.05) and the high value of adjusted R-squared (approximately 83%) shows that the two independent / explanatory variables are able to explain 83% of the variance in the dependent variable. This proves that the above regression model is good enough for interpretation and prediction. As part of interpretation it can be stated (Supplementary Fig. 4) that a 1unit increase (when the concentration of Dye is unchanged) in concentration of Cr III decreases the Biomass concentration by 0.016 units. Also it can be stated that a 1unit increase (when the concentration of Cr III is unchanged) in concentration of Dye decreases the Biomass concentration by 0.017 units. Fig. 2 shows the histogram for checking normality assumption in regression analysis. From the histogram it is clear that the error terms are approximately normally distributed. Further the results of Shapiro-Wilk test confirm that the error terms follow normal distribution (Supplementary Fig. 5). The low values of VIF (less than 5) show that there is no multi-collinearity between the two independent variables. The Durbin Watson test results demonstrate that the error terms are serially uncorrelated. Finally, the Breusch-Pagan test proves that there is no heteroscedasticity. Hence the above interpretations hold good and the multiple linear regression model can be used for predictions also. From the statistical validation it may be concluded that both “concentration of Cr III” and “concentration of Dye” have significant impacts on the “Biomass Concentration”.
4. CONCLUSION
Growing cells of A. tamarii was observed to be potent for the removal of color and chromium from synthetic solution of chromium, RR120 dye and binary system of both. Bioremediation of the dye was visualized by SEM analysis. A. tamarii, therefore, may be considered to have the potential to biologically treat industrial waste-water contaminated with dye as well as chromium. Also, the proposed bioremediation process is more environment friendly than other conventional processes. The fungi showed higher tolerance towards dyes/metals than other micro-organisms and higher potential to remove pollutant. The validation study done using multiple linear regression model (run using R&R studio) proved that the regression model (adjusted R2 is 83%) is good enough for interpretation and prediction. Further the statistical model also corroborates the fact that the concentration of dye as well as chromium significantly impacts the biomass concentration.
References
-
Akar, T., Tunali, S. (2006) Biosorption characteristics of Aspergillus flavus biomass for removal of Pb(II) and Cu(II) ions from an aqueous solution. Bioresource Technology 97(15), 1780-1787
[https://doi.org/10.1016/j.biortech.2005.09.009]

-
Bachate, S.P., Nandre, V.S., Ghatpande, N.S., Kodam, K.M. (2013) Simultaneous reduction of Cr(VI) and oxidation of As(III) by Bacillus firmus TE7 isolated from tannery effluent. Chemosphere 90(8), 2273-2278
[https://doi.org/10.1016/j.chemosphere.2012.10.081]

-
Baruthio, F. (1992) Toxic effects of chromium and its compounds. Biological Trace Element Research 32(1-3), 145-153.
[https://doi.org/10.1007/BF02784599]

-
Bertolo, R., Bourotte, C., Hirata, R., Marcolan, L., Sracek, O. (2011) Geochemistry of natural chromium occurrence in a sandstone aquifer in Bauru Basin, São Paulo State, Brazil. Applied Geochemistry 26(8), 1353-1363
[https://doi.org/10.1016/j.apgeochem.2011.05.009]

-
Fomina, M., Charnock, J.M., Hillier, S., Alvarez, R., Gadd, G.M. (2007) Fungal transformations of uranium oxides. Environmental Microbiology 9(7), 1696-1710
[https://doi.org/10.1111/j.1462-2920.2007.01288.x]

-
Ghosh, A., Dastidar, M.G., Sreekrishnan, T.R. (2017) Response surface modeling of bioremediation of acid black 52 dye using Aspergillus flavus. Water Science and Technology 75(12), 2864-2874
[https://doi.org/10.2166/wst.2017.167]

-
Ghosh, A., Dastidar, M.G., Sreekrishnan, T.R. (2017) Response surface optimization of bioremediation of Acid black 52 (Cr complex dye) using Aspergillus tamarii. Environmental Technology 38(3), 326-336
[https://doi.org/10.1080/09593330.2016.1192225]

-
Ghosh, A., Ghosh Dastidar, M., Sreekrishnan, T.R. (2017) Bioremediation of Cr complex dyes and treatment of sludge generated during the process. International Biodeterioration & Biodegradation 119, 448-460
[https://doi.org/10.1016/j.ibiod.2016.08.013]

-
Ghosh, A., Ghosh Dastidar, M., Sreekrishnan, T.R. (2016) Bioremediation of a Chromium Complex Dye (Navilan yellow RL) using Aspergillus flavus and Aspergillus tamarii. Chemical Engineering & Technology 39(9), 1636-1644
[https://doi.org/10.1002/ceat.201500515]

-
Ghosh, A., Ghosh Dastidar, M., Sreekrishnan, T.R. (2015) Recent Advances in Bioremediation of Heavy Metals and Metal Complex Dyes: Review. Journal of Environmental Engineering C4015003, 142(9), 1-14
[https://doi.org/10.1061/(ASCE)EE.1943-7870.0000965]

-
Ghosh, A., Dastidar, M.G., Sreekrishnan, T.R. (2014) Biosorption and Biodegradation of Chromium Complex Dye Using Aspergillus Species. Journal of Hazardous, Toxic, and Radioactive Waste 18(4), 1-9
[https://doi.org/10.1061/(ASCE)HZ.2153-5515.0000230]

-
Guimaraes-Soares, L., Felicia, H., Bebianno, M.J., Cassio, F. (2006) Metal-binding proteins and peptides in the aquatic fungi Fontanospora fusiramosa and Flagellopora curta exposed to severe metal stress. Science of the Total Environment 372, 148-156
[https://doi.org/10.1016/j.scitotenv.2006.09.017]

- Gujarati, D.N. (2009) Basic econometrics. Tata McGraw-Hill Education.
-
Horsfall, M., Ogban, F., Akporhonor, E.E. (2006) Sorption of chromium (VI) from aqueous solution by cassava (Manihot sculenta Cranz.) waste biomass. Chemistry and Biodiversity 3(2), 161-174
[https://doi.org/10.1002/cbdv.200690019]

-
Mills, C.T., Morrison, J.M., Goldhaber, M.B., Ellefsen, K.J. (2011) Chromium (VI) generation in vadose zone soils and alluvial sediments of the southwestern Sacramento Valley, California: a potential source of geogenic Cr (VI) to groundwater. Applied Geochemistry 26(8), 1488-1501
[https://doi.org/10.1016/j.apgeochem.2011.05.023]

-
Miretzky, P., Cirelli, A.F. (2010) Cr (VI) and Cr (III) removal from aqueous solution by raw and modified lignocellulosic materials: a review. Journal of Hazardous Materials 180(1-3), 1-19
[https://doi.org/10.1016/j.jhazmat.2010.04.060]

-
Mishra, A., Malik, A. (2014) Metal and dye removal using fungal consortium from mixed waste stream: optimization and validation. Ecological Engineering 69, 226-231
[https://doi.org/10.1016/j.ecoleng.2014.04.007]

- Pubchem NCBI website: https://pubchem.ncbi.nlm.nih.gov/compound/Reactive-Red-120, .
- Pubchem NCBI website: https://pubchem.ncbi.nlm.nih.gov/compound/C.I.-Acid-Blue-158_-disodium-salt, .
-
Puyen, Z.M., Villagrasa, E., Maldonado, J., Diestra, E., Esteve, I., Solé, A. (2012) Biosorption of lead and copper by heavy-metal tolerant Micrococcus luteus DE2008. Bioresource Technology 126, 233-237
[https://doi.org/10.1016/j.biortech.2012.09.036]

-
Rakhshaee, R. (2011) Rule of Fe0 nano-particles and biopolymer structures in kinds of the connected pairs to remove Acid Yellow 17 from aqueous solution: Simultaneous removal of dye in two paths and by four mechanisms. Journal of Hazardous Materials 197, 144-152
[https://doi.org/10.1016/j.jhazmat.2011.09.067]

-
Ranjusha, V.P., Pundir, R., Kumar, K., Dastidar, M.G., Sreekrishnan, T.R. (2010) Biosorption of Remazol Black B dye (Azo dye) by the growing Aspergillus flavus. Journal of Environmental Science and Health Part A 45(10), 1256-1263
[https://doi.org/10.1080/10934529.2010.493812]

-
Sari, A., Tuzen, M. (2008) Biosorption of total chromium from aqueous solution by red algae (Ceramium virgatum): equilibrium, kinetic and thermodynamic studies. Journal of Hazardous Materials 160(2), 349-355
[https://doi.org/10.1016/j.jhazmat.2008.03.005]

-
Saroj, S., Kumar, K., Pareek, N., Prasad, R., Singh, R.P. (2014) Biodegradation of azo dyes Acid Red 183, Direct Blue 15 and Direct Red 75 by the isolate Penicillium oxalicum SAR-3. Chemosphere 107, 240-248
[https://doi.org/10.1016/j.chemosphere.2013.12.049]

-
Sen, M., Dastidar, M.G., Roychoudhury, P.K. (2005) Biosorption of chromium(VI) by nonliving Fusarium sp. isolated from soil. Practice Periodical of Hazardous, Toxic, and Radioactive Waste Management 9(3), 147-151
[https://doi.org/10.1061/(ASCE)1090-025X(2005)9:3(147)]

-
Sen, M., Dastidar, M.G., Roychoudhury, P.K. (2007) Biological removal of Cr(VI) using Fusarium solani in batch and continuous modes of operation. Enzyme and Microbial Technology 41(1), 51-56
[https://doi.org/10.1016/j.enzmictec.2006.11.021]

- Sen, M., Dastidar, M.G. (2011) Biosorption of Cr (VI) by resting cells of Fusarium solani. Iranian Journal of Environmental Health Science & Engineering 8(2), 153-158.
-
Skorik, Y.A., Pestov, A.V., Yatluk, Y.G. (2010) Evaluation of various chitin-glucan derivatives from Aspergillus niger as transition metal adsorbents. Bioresource Technology 101(6), 1769-1775
[https://doi.org/10.1016/j.biortech.2009.10.033]

-
Sun, J., Shao, Z. (2007) Biosorption and bioaccumulation of lead by Penicillium sp. Psf-2 isolated from the deep sea sediment of the Pacific Ocean. Extremophiles 11(6), 853-858.
[https://doi.org/10.1007/s00792-007-0097-7]

- Sullivan, R.J. (1969) Air pollution aspects of chromium and its compounds.
-
Wu, X., Hui, K.N., Hui, K.S., Lee, S.K., Zhou, W., Chen, R., Hwang, D.H., Cho, Y.R., Son, Y.G. (2012) Adsorption of basic yellow 87 from aqueous solution onto two different mesoporous adsorbents. Chemical Engineering Journal 180, 91-98
[https://doi.org/10.1016/j.cej.2011.11.009]

-
Zhang, J., Yang, K., Wang, H., Lv, B., Ma, F. (2016) Biosorption of copper and nickel ions using Pseudomonas sp. in single and binary metal systems. Desalination and Water Treatment 57(6), 2799-2808
[https://doi.org/10.1080/19443994.2014.983177]

Appendix
SUPPLEMENTARY MATERIALS

SEM micrograph of the fungi cells after bioremediation of RR120 dye and chromium in the ratio of 92:8.6 mg/L, respectively.

SEM micrograph of the fungi cells after bioremediation of RR120 dye and chromium in the ratio of 50:50 mg/L, respectively.