Changes in Inorganic Chemical Species in Fog Water over Delhi
Copyright © 2022 by Asian Association for Atmospheric Environment
This is an open-access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Heavy fogs occur during the winter period over the part of northern India and impact aviation, public transport, the economy, public life, etc. During winter, fog water (FW) and non-monsoonal rainwater (NMRW) samples were collected in Delhi, which is a highly polluted and populated megacity in northern India. The collected FW and NMRW samples were analyzed for their inorganic chemical constituents (F-, Cl-, SO42-, NO3-, NH4+, Na+, K+, Ca2+, and Mg2+). The volume-weighted mean (VWM) pH, conductivity, and total dissolved solids (TDS) of FW were 6.89, 206 μS cm-1, and 107 mg L-1, respectively, indicating the dominance of alkaline species. The total measured ionic constituents (TMIC) in FW and NMRW were 5,738 and 814 μeq L-1, respectively, indicating highly concentrated FW in Delhi. The TMIC in FW were factors of 16 and 7 times more concentrated than MRW and NMRW samples, respectively. The concentrations of inorganic acidic species (SO42- and NO3-) in FW were much higher than in monsoon rainwater (MRW: 3 and 5 times) and NMRW (8 and 12 times), respectively. Also, the concentrations of SO42- and NO3 in NMRW were approximately double compared to MRW indicating higher acidic species concentrations during the winter season over Delhi region. Significant decadal growth in the mean concentrations of ionic species in FW (SO42- - ~9 times; NH4+ - double) were observed between 1985 and 2010. However, the nitrate decreased by ~28%. The higher SO42- is likely from heavy-duty vehicles that burn sulfur-containing fuel. The anions in FW, MRW, and NMRW contributed 20, 42, and 43%. However, the cation contributions were 80, 58, and 57%, respectively. The anion contributions were lower in FW than MRW and NMRW indicating the weak formation of acidic species in fog water. The observed alkalinity suggests that it is unlikely for acid precipitation to be present in this region.
Keywords:
Fog water, Rainwater chemistry, Secondary aerosol species, Acidity, Alkalinity1. INTRODUCTION
Fog is a near-ground cloud and is categorized as cold or warm. Fogs have significant impacts on regional air quality, visibility (less than 1,000 meters) (Tiwari et al., 2012, 2011; Niu et al., 2010), traffic systems, human health, the economy (Gultepe et al., 2007), and the ecosystem (Shigihara et al., 2009; Butler and Trumble, 2008; Jacobson, 1984). Generally, fog formation occurs in the lower layer of the troposphere (up to 300 meters above the ground) where the particles and gaseous pollutants are abundant. Because the fog droplets are smaller than rain droplets (~100 times), constituents in fog droplets will be more highly concentrated (Lange et al., 2003). The physical and chemical characteristics of fog water and cloud water have been studied around the world because droplets can incorporate particulate and gaseous species and transform them through aqueous-phase reactions (Simon et al., 2016; Wang et al., 2015; Gilardoni, 2014; Watanabe et al., 2011, 2006; Pruppecher and Klett, 2010; Raja et al., 2009; Collett et al., 2008, 2001; Fukuzaki et al., 2002; Igawa et al., 2001; Jacob et al., 1987; Walter, 1983). Langner and Rodhe (1991) suggested that more than 80% of the atmospheric sulfate (secondary aerosol) is produced from SO2 (gas) in cloud reactions. Pandis et al. (1990) reported that fog droplets scavenged water-soluble gaseous species such as nitric acid and ammonia. Scavenging of atmospheric particles and gaseous species by the fog determines the ionic compositions of the fog water (Sickles et al., 1999). Fog water compositions have been measured in diverse environments around the world such that the observational database of fog water compositions has increased (Herckes et al., 2015; Li et al., 2011). However, in some locations, only a few studies have been reported.
The Indo-Gangetic Plains (IGP) of India experiences intense fog conditions during the winter season. Acid fogs have been reported in the United States, Europe, Japan, Taiwan, etc. (Simon et al., 2016; Igawa et al., 2001; Watanabe et al., 2001; Jacob et al., 1987). In the northern part of India, fog occurs during the winter period when temperatures reach minimum values (<5.0°C) following Western Disturbances (WDs). The WD is a weather phenomenon causing rain and fog over this region during the winter period. It is extra-tropical storms originating in the Mediterranean region that brings sudden winter rains to the northwestern parts of the Indian subcontinent (Jenamani and Rathore, 2015). The WDs bring a lot of moisture causing precipitation (rain fog, snow, etc.) over the northern part of India. In addition, irrigation for winter crops and the irrigation network in this region adds a significant amount of moisture to the lower atmosphere (Badarinath et al., 2009). Some studies since the 1980s have been made during intense fog periods over Delhi (Ali et al., 2004; Kapoor et al., 1993; Khemani et al., 1987) and their results showed that the ionic composition of fog water was mostly alkaline.
Fog water and rainwater were previously studied when polluting industries began to move from residential areas, fuels for commercial vehicles were being changed (introduced compressed natural gas) and metro-rail infrastructure was instituted in Delhi (Ali et al., 2004). A decade after the adoption of new strategies to improve air quality, the Delhi and National Capital Region still experienced substantial air pollution during winter (foggy episodes) including violation of the National Ambient Air Quality Standards for air pollutants (Tiwari et al. 2016a, 2016b, 2015; Chate et al., 2014). This study of the ionic composition of fog water collected in 2010 was to assess the impacts of changing emissions from a decadal growth in population, the 200% increase in vehicles plying the roads, and their associated emissions (Sahu et al., 2015, 2011; Nagpure et al., 2013), and the infrastructural developments made for the Commonwealth Games of 2010 held in Delhi.
The present study investigated the ionic composition of fog water (FW) and rainwater (RW) in Delhi during the winter (non-monsoonal) period and compared the results with those reported from previous studies at this site to quantify the decadal changes in chemical species. The concentrations of primary and secondary chemical species in fog water were measured during the foggy events. The present study has four major objectives: (i) to characterize the chemistry of FW and RW during the winter (cold) period, (ii) to provide a better understanding of secondary aerosols (sulfate: SO42- and nitrate: NO3-) concentrations in FW and RW, (iii) to identify the source regions of the measured ionic constituents of FW and RW, and (iv) to assess the decadal changes of measured ions especially secondary species in FW at a highly polluted site in IGP region.
2. STUDY AREA AND METEOROLOGICAL CONDITIONS DURING FOGGY PERIOD
Delhi and its surrounding region called the National Capital Region (NCR) of Delhi is India’s largest agglomeration and is one of the largest in the world concerning population and pollution. The population of Delhi was more than 13.8 million inhabitants (population census of 2001) in 2001 and has increased to approximately 16.7 million in 2010 (Census of India, 2011), exhibiting a decadal growth of about 21%. The urbanization of Delhi and its surrounding region have put great stress on the local environment with one of its effects being fog-related disasters that happen frequently in the winter.
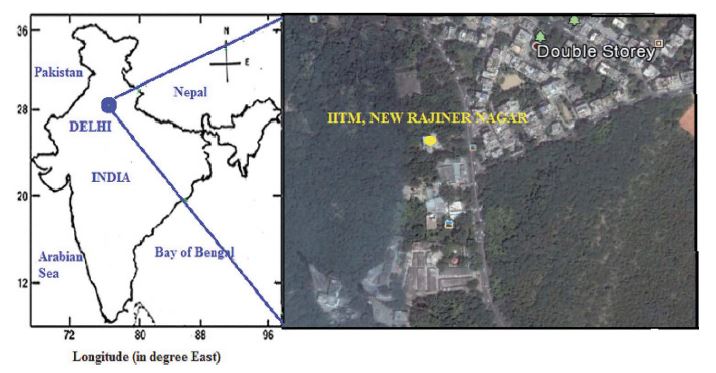
Sampling location of the fog water and rainwater in Delhi (28.62°N, 77.17°E: 217 m asl). The local map area is around 2×2 km.
The FW and RW sampling site were on the premises of the Indian Institute of Tropical Meteorology (IITM) in Delhi (28.62°N, 77.17°E, 217 m above meter sea level). The site is a relatively open area to the East, South, and North with a greenbelt on these three sides. There are residential buildings towards the west at a distance of 100 m as described elsewhere (Tiwari et al., 2015, 2011). A major road lies to the west and a major link road led to the Gurgaon, Haryana is on the east side (Tiwari et al., 2015). There is heavier traffic in the morning and evening hours. It has a semi-arid climate and is located between the rain-washed Indo-Gangetic plains (IGP), one of the highly polluted and populated regions in the world, and the semi-arid tract of Rajasthan to the east and southwest, respectively. The city (~18 million inhabitants) is ~1,100 km away from the Arabian Sea (Chelani et al., 2012). The major particulate matter (PM) sources in Delhi are small/large industries, brick kilns, light, and heavy-duty vehicles mostly operated by diesel fuel, and three coal-fired power plants (Srivastava and Jain, 2008). There is also a background of PM transported into Delhi from the IGP and Desert region.
Meteorological data such as wind speed (WS) and direction (WD), temperature (T), dew point temperature (DPT) were measured by an automatic weather station at IITM New Delhi (Bisht et al., 2016). Visibility was measured with a Drishti Transmissometer at the Indira Gandhi International Airport which is 12 km (aerial) away from the sampling site in the West direction (Mohan et al., 2015). During January 2010, the mean WS, T, DPT, and visibility were 1.45±1.3 m s-1, 12.7±4.3°C, 9.8±2.0°C and 720±415 m, respectively. The wind rose based on the observed wind direction data measured in January 2010 is shown in Fig. 2. The resultant wind vector was north (342°: ~67%) with a mean wind speed of 2.1 m s-1. Higher WS were observed when the WD was from the west contributing 31%. Calm conditions (below 0.5 m s-1) in January were observed for ~22% of the time.
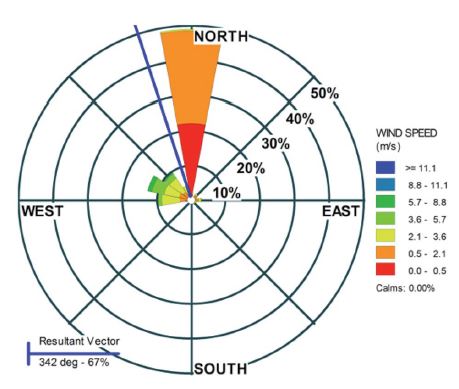
Distribution of wind speed (m s-1) with wind direction (degree) at sampling site during January 2010.
2. 1 Fog Water and Rainwater Sampling during the Winter Season
The FW (12nos) and NMRW (2nos) samples were collected using an active fog water collector and a bulk rain collector during January 2010 and November 2010, respectively. The fog sampler maintained a temperature of -15°C inside a stainless-steel cooling chamber with dimensions of 10×10×22 cm3 and was used to collect fog droplets by impaction. Ambient air was pulled at 0.2 m3 min-1 through the chamber where it cooled until the ice was formed. The sampler was operated for a sufficient period to ensure that an adequate sample volume was collected for chemical analysis. The collector was then switched to the melting mode and the liquid sample decanted. Details on sampling technique (equipment and method of sample collection and preservation) can be found elsewhere (Ali et al., 2004; Khemani et al., 1987). The cooling chamber of the equipment was thoroughly washed several times with de-ionized water before and after each fog sampling. The pH of the rinse water was checked. NMRW samples were collected with a bulk collector that consists of a high-quality polypropylene funnel having a diameter of 20 cm connected to a plastic bottle (2 liters). It was placed on the rooftop of the Institute building (~15 m above the ground level) (Tiwari et al., 2016a). Thymol (<5 mg) was added to the FW and NMRW samples as a preservative to prevent significant changes in the concentrations via biological degradation before transferring the samples in bottles (Gillett and Ayers, 1991). The samples were stored in a 4°C refrigerator until the chemical analysis.
The pH and TDS of each sample were measured with a digital pH meter (Cyber Scan 510 pH meter), however, the conductivity (Elico CM-183) meter was used for the measurement of electric conductivity. During pH measurement, KCl reference and glass electrodes were standardized with reference solutions with pH values of 4.1 and 9.2. The sample volumes were between 27.7 and 73.8 mL for FW and 2 to 3 mm for RW. The samples were analyzed for anions (F-, Cl-, SO42-, and NO3-) and cations (Na+, K+, Ca2+, and Mg2+) using ion chromatography (Dionex-100, USA) and atomic absorption spectrophotometry (Perkin-Elmer, Model 373), respectively (Budhavant et al., 2014). The detection limit of the measured anions (Cl-, NO3-, and SO42-) were 3.38, 4.35 and 7.29 μeq L-1 and for cations (Na+, K+, Ca2+, and Mg2+) were, 0.43, 0.26, 1.00 and 0.42 μeq L-1, respectively (Bisht et al., 2016; Tiwari et al., 2016a). Ammonium ion (NH4+) concentration was measured by the colorimetric Indophenol Blue method with a UV/Visible double beam spectrophotometer (Bausch and Lomb, USA) (Weatherburn, 1967). The concentrations of HCO3- were estimated from the pH values using the following relationship described by Kulshrestha et al. (2003):
| (1) |
The free acidity (H+) was also calculated from the measured pH (Tiwari et al., 2012). Blank samples were collected by spraying de-ionized water into the fog and rain collectors before and after during the campaign period to assess any contamination. The blank samples were analyzed in the same manner as the FW and RW. Small blank concentrations were obtained and the blank concentrations of individual species were subtracted from the sample values. Quality checks were performed by calculating the electro-neutrality balance of chemical species, and by the ionic balance. In the case of ionic balance, the ratios of the sum of anions to the sum of cations were estimated and high variability was observed indicating that several organic acids that were not measured (Raja et al., 2009; Collett et al., 2008). Samples collected in the winter when Delhi was highly affected by aged biomass burning aerosol coming from agricultural stubble burning in the Indo-Gangetic Plains would be strongly affected by the high levels of particulate matter pollution. Prior studies (Fang et al., 2021; Deshmukh et al., 2019; Cong et al., 2015; Falkovich et al., 2005) have reported high concentrations of mono and di- organic acids in biomass burning aerosol that could be scavenged by the rain or fog. The filters used to filter the samples after filtration were found in black color indicating high organic PM scavenging due to biomass burnig. Around 17.66% organic acids (formic acid, acetic acid, and oxalic acid) were found in cloud water and rainwater at Mount Lu, a mountain site located in the acid rain-affected area of South China, from August to September of 2011 and March to May of 2012 (Sun et al., 2016).
2. 2 Estimation of Neutralization Factors
The neutralization factors (NFs) are a measure of the effectiveness of individual cations (Ca2+, Mg2+, K+, and NH4+) on the neutralization of atmospheric acidity. The NFs in FW and NMRW were computed for different alkaline constituents (μeq L-1) using the following formula
| (2) |
Where X is specific alkaline species such as Ca2+, Mg2+, K+, and NH4+. Generally, the specific chemical species and sulfate were used as their non-sea salt (nss) contributions in the estimation of NFs. However, in this case, concentrations were used directly because Delhi is not strongly affected by marine sources. The origin of FW and NMRW during the winter is due to western disturbances, a natural phenomenon that occurs in the winter season. They are non-monsoonal precipitation patterns driven by the westerly winds. Under such atmospheric conditions, higher atmospheric pollutant concentrations were observed as a result of lower ventilation and poorer mixing of the pollutants (Ali et al., 2004). In the estimation of NF by alkaline components, nss fractions were considered negligible.
3. RESULTS AND DISCUSSION
3. 1 Variability of pH in FW and NMRW in Delhi
The chemical species enter into the atmosphere from a variety of anthropogenic and natural sources. Acids (such as sulfuric and nitric) are formed by the oxidation of SO2 and NOx emitted by the combustion of fossil fuel by industrial activities and motor vehicles. Particulate alkaline species (calcium and magnesium) are present from the aerosolization of calcareous soils. Ammonia is emitted by both biogenic and anthropogenic sources. Atmospheric CO2 concentrations have increased from the beginning of the industrial era until now from the combustion of fossil fuels (coal, oil, natural gas, etc.). The level of CO2 in 2010 was ~390 ppm (scrippsco2.ucsd.edu/, www.esrl.noaa.gov/gmd/ccgg/trends/, and IPCC, 2014) that will affect the pH of the precipitation. Charlson and Rodhe (1982) suggested that a threshold value (pH=5.61) defines “acid rain” based on the atmospheric equilibrium with CO2. The volume-weighted mean (VWM) pH of FW and NMRW were 6.89 and 7.18, respectively. These values are more alkaline than water in equilibrium with atmospheric CO2. The increased pH levels are due to the high concentrations of alkaline species (Ca2+, Mg2+, K+, and NH4+) that were present in the fog or rain. Khemani et al. (1987) found high pH values in 1985 (6.4 to 7.6) in fog water at the same location due to the dominance of alkaline species. One and half decades later (in 2001/2002) at the same location, Ali et al. (2004) reported pH values of 6.66 and 7.04 in FW and NMRW, respectively. They also suggested high concentrations of alkaline species. Recently, Tiwari et al. (2016a) studied monsoonal rainwater chemistry over the highly polluted, densely populated IGP region from 2009 to 2011 by analyzing 687 samples collected over three consecutive summer monsoon seasons and reported lower mean pH value (5.73±0.17) for this monsoon rainwater (MRW) period with 16% acidic RW (pH< 5.61). They also reported higher/lower conductivity (33.6 μS cm-1/20.6 μS cm-1) during alkaline/acidic rainy events. The conductivity and TDS were also measured in FW and were found to be higher (mean: 206±153 μS cm-1) and (107±79 mg L-1) than that of the IGP region.
3. 2 Chemistry of Fog Water and Rainwater during Winter Period in Delhi
The water (fog or rain) volumes influence the concentrations of their alkaline and acidic constituents in MRW (Tiwari et al., 2016a) and non-monsoon rainwater: NMRW (present study). The total mean ionic constituent (TMIC) in FW, MRW, and NMRW were 5738, 360, and 814 μeq L-1, respectively, indicating that highly concentrated fog occurs over the northern part of India. The TMIC in FW were factors of 16 and 7 times more concentrated than MRW and NMRW samples, respectively. The higher TMIC in FW and NMRW during the cold season (from November to January) compared to the monsoon season (July to September) is due to lower boundary layer height during the colder period (Wang and Li, 1994). In this study, volume-weighted mean measured ionic concentrations of F-, Cl-, SO42-, NO3-, HCO3-, Na+, K+, Ca2+, Mg2+, and NH4+ in FW, MRW (Tiwari et al., 2016a), and NMRW over Delhi during 2010 are presented in Fig. 3. The ionic contribution in FW was in the order of Ca2+, SO42-, Na+, K+, Mg2+, NH4+, NO3-, HCO3-, Cl-, and F-. In NMRW, they were slightly different from fog water having the highest contribution species was Ca2+ followed by HCO3-, SO42-, Mg2+, NH4+, NO3-, Cl-, Na+, K+, and F-. In MRW, the trend was similar to NMRW except for a similar molar ratio (1.58) of sea salt constituent indicating that most of the sea salt arrived from marine sources. The concentrations of acidic species (SO42- and NO3-) in FW were much higher than that MRW (3 and 5 times) and NMRW (8 and 12 times), respectively. The concentrations of SO42- and NO3- were around a double in NMRW compared to MRW indicating the higher acidic species concentrations during the winter season. The relatively higher concentrations of secondary aerosols species (SO42- and NO3-) in Delhi were likely the result of the combustion of fossil fuels from thermal power plants, refineries, other industrial activities, diesel vehicles etc.
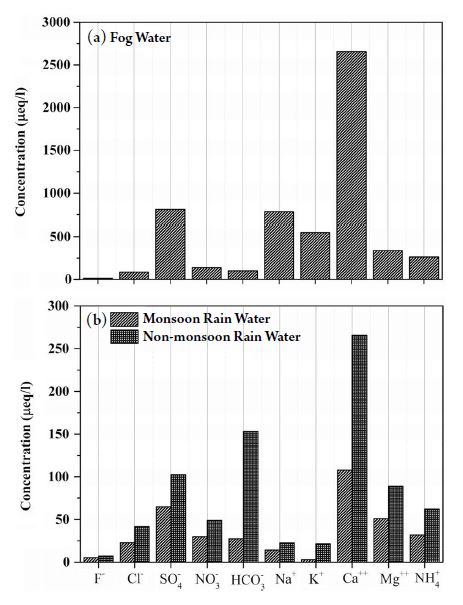
The mean ionic concentrations of F-, Cl-, SO42-, NO3-, HCO3-, Na+, K+, Ca2+, Mg2+, and NH4+ of fog water (during the foggy period) and rainwater (monsoon and non-monsoon period) over Delhi during 2010.
In Delhi, there are three thermal power plants (Badarpur Thermal Power Station, Rajghat Power House Station, Pragati Power Station) located 20 km southeast of the sampling site on the banks of the Yamuna River. Several other thermal power plants (Faridabad, Panipat, Dadri, etc) are located within a 200 km radius of Delhi. Earlier studies reported that the higher concentrations of SO42- in the fog was the result of the rapid heterogeneous conversion of SOx to SO42- (Ali et al., 2004; Munger et al., 1983; Sander and Seinfeld, 1976). Sulfur dioxide, which is emitted by diesel engines, given the relatively high sulfur content in Indian diesel fuels that were being phased out at the time of the sampling. Jain et al. (2000) reported a significant fraction of nss SO42- in RW over Delhi. The switch to natural gas to power motor vehicles will result in high NO emissions that can also be heterogeneously oxidized resulting in high nitrate concentrations. The concentrations of Ca2+ in FW were observed much higher than that MRW (25 times) and NMRW (10 times). However, in the comparison between NMRW and MRW, Ca2+ was around 2.5 times higher in NMRW than MRW. This higher concentration of Ca2+ in FW and NMRW may be the result of the re-suspension of calcareous soil. It could be associated with other sources such as suspension from paved and unpaved roads (Sahu et al., 2011). Khemani et al. (1989) reported that all the northern parts of India including Delhi are affected by natural dust particles, blown by winds from the arid Thar Desert located to the northwest. Enhanced constructional activities using concrete in Delhi and its surroundings may also be responsible for the drastic increase in the Ca2+ concentrations.
In FW, the concentrations of ammonium were 4 and 8 times higher than MRW and NMRW, respectively. In addition, the non monsoonal ammonium mean concentration was approximately double compared to the monsoon period. Earlier studies (Ali et al., 2004; Kapoor et al., 1993) suggested that the major source of NH3 emissions is from urine excretion, fertilizer use, etc. over this region. During the study period, the potassium (K+) concentrations were substantially higher than that NMRW and MRW and were due to biomass burning in this region. In NMRW, the K+ values were around seven times higher than in MRW. Kaskaoutis et al. (2014) reported large-scale crop residue burning was observed over the northwestern part of India during the post-monsoon and winter periods. Several studies indicated the major source of potassium was from biomass burning (Perrino et al., 2014; Pachon et al., 2013 and references therein). Lu et al. (2010) suggested that biomass burning is the major source of K+ in fog water.
Sea salt (Cl- and Na+) components were found to have large variability in fog water compared to NMRW and MRW. However, total sea salt constituents were low given the distances to the nearest oceans and are more likely to come from entrained desert dust from areas like Dasht-e Kavir and the Great Salt Desert in Iran and the Rann of Kutch, a salt marsh in the Thar Desert. The chloride was around 4 and 2 times higher in FW than that of MRW and NMRW, whereas, the contribution of sodium was large in fog water and was ~54 and 35 times higher than MRW and NMRW, respectively (Senaratne et al., 2005). This large variability is due to the entrainment of anthropogenic aerosol during the winter period. The fractional contributions of individually measured ions in FW, MRW, and NMRW in Delhi during 2010 were estimated (Fig. 4) and indicate the entrainment of soils chemical species are higher in FW than RW. The contribution of anions, and cations in FW, MRW, and NMRW were 20%, 42%, and 43%, and 80%, 58%, and 57%, respectively. The contribution of anions was much lower in FW than MRW and NMRW indicating weaker scavenging of anions by fog. This difference may be due to the height of fog formation. Fog forms near the ground level (up to 300 meters), whereas rainwater forms in the lower troposphere (up to 3 km), and the cloud droplets persist over longer times, permitting the build-up of oxidized gaseous species.
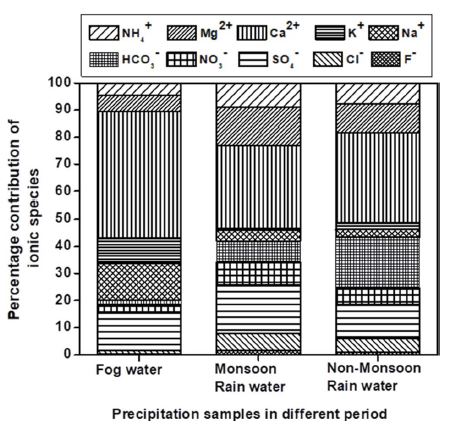
Percentage contribution of major ions in fog water, rainwater in monsoon, and the non-monsoon period over Delhi during 2010.
The ratios of measured ions were estimated to identify the relative contributions of sulphuric and nitric acids to FW and RW over Delhi. The average ratios of H+/(NO3-+SO42-) in FW, MRW, and NMRW were 2.8×10-5, 7×10-4, and 1×10-4, respectively, indicating that almost all the acidity was neutralized in FW, MRW, and NMRW. The ratios ([Ca2+]+[K+]+[Mg2+]+[NH4+])/([NO3-]+[SO42-]) values in FW, MRW and NMRW were greater than unity (4.8, 2.2, and 3.0) indicating that K+, Mg2+, NH4+ and Ca2+ ions play a major role in neutralizing the acidity in the FW, MRW, and NMRW, respectively. The ratios of NH4+/NO3- (1.9, 1.08, and 1.27) and NH4+/SO42- (0.32, 0.49 and 0.61) in FW, MRW, and NMRW, respectively, were calculated. These results indicate that ammonium nitrate (NH4NO3) may be predominant over ammonium sulfate [(NH4)2SO4] in fog water (Seinfeld and Pandis, 2006). The dominant ionic composition in FW was compared with the ionic composition of FW collected in China (at Pancheng in the Nanjing area of China) during December 2006 and December 2007 (Lu et al., 2010). The sum of anions and the sum of cations in the present study are compared to those in China in Table 1. The sum of the acidic and alkaline species was much lower than in China. The fog water in the present study and China are alkaline (mean pH: 6.89), respectively. In contrast, acid fogs have been observed in many urban settings (Waldman et al., 1982). The high pH values in India and China are due to the presence of alkaline components. However, in the US, there is high nitrate but low concentrations of alkaline components in the fog water. The present study indicated that the impact of gaseous pollutants emanating from local and regional sources is less than that of suspended soil.
3. 3 Decadal Changes in Ionic Composition in the Fog Water
The present study was compared with fog water chemistry studies conducted at the same location in 1985 (Khemani et al., 1986) and 2001/2002 (Ali et al., 2004). The results are summarized in Table 2. The pH values during 1985, 2001/2002, and 2010 were 7.15, 5.81, and 6.89, respectively and it was observed that the pH has decreased (23%) from 1985 to 2001/2002. However, from 2001/2002 to 2010, the pH increased (18%). Between 1985 and 2010, there was only a 4% reduction of pH in FW. The sum of the average measured ionic constituent was 5,738 μeq L-1 in 2010, which is approximately a factor of three (2,040 μeq L-1) and two (3,050 μeq L-1) higher than the 2001/2002 and 1985 values, respectively. The sum of measured anions and cations in FW was 660, 915, and 1,149 μeq L-1, and 2,390, 1,125, and 4,590 μeq L-1 during 1985, 2001/2002, and 2010, respectively. Large decadal changes in the concentrations of anions and cations in FW were due to the implementation of different strategies to reduce pollution levels in Delhi and its surroundings. For anions, the concentrations increased approximately 72% in the two and half decades (1985 to 2010), 38% in one decade (from 2001/2002 to 2010), and only 25% from 1985 to 2001/2002. However, cations increased 92% in two and half decades (1985 to 2010) and a factor of three in one decade (from 2001/2002 to 2010). The cations concentrations decreased drastically (53%) in one and half decades (from 1985 to 2001/2002). Significant decadal growth in the mean concentrations of secondary aerosols in FW (SO42- - about 9 times; NH4+ - double) was observed from 1985 to 2010. Nitrate concentrations were reduced by about 28%. This reduction was due to the stringent implementation of fuel quality upgrades, vehicular emission limits, better maintenance of engines, etc. The reduction in sulfate from 2001 onward can also be linked to the conversion of all commercial goods and passenger vehicles to CNG engines. However, SO42- values increased afterward because of the extensive use of coal in power plants and brick kilns. Aerosolized soil (calcium), as well as the biomass burning marker potassium, approximately doubled and tripled from 1985 to 2010, respectively. However, there were no changes in the concentrations of magnesium. From 1985 to 2001/2002, the concentrations of sulfate and ammonium increased around six and three-fold, respectively. However, chloride, nitrate, sodium, potassium, calcium, and magnesium drastically declined by 40, 62, 74, 88, 64, and 76%, respectively. The decadal population growth (around three times from 1985 (6,770,000 in 1985) to 2010 (16,349,831 in 2011) in Delhi (Census of India, 2011), number of vehicles, anthropogenic activities, construction of infrastructure associated with changes in land useland cover across Delhi, etc. (Sahu et al., 2015, 2011; Nagpur et al., 2013) are likely to be responsible for the increases in the anions (dominantly SO42-) and cations (predominantly Ca2+) in FW sampled during 2010 compared to 1985. The use of commercial vehicles in Delhi is increasing at an average rate of 7 percent annually. They are mostly diesel vehicles that emit large quantities of sulfur into the regional atmosphere (Das et al., 2010).
3. 4 Estimation of Neutralization Factors
The acidic or alkaline nature of any precipitation is determined by estimating the potential acid-neutralizing capacity of alkaline components (Ca2+, Mg2+, K+, and NH4+) that are present in the atmosphere from natural sources such as soils (Srinivas et al., 2001) except ammonium which is released from anthropogenic activities and urine excretion (Kapoor et al., 1993). Neutralization factors (NFs) for Ca2+, Mg2+, K+, and NH4+ identify the role of alkaline species in neutralizing the acidic components in the atmosphere (Srinivas et al., 2001). The average NFs for Ca2+, Mg2+, K+, and NH4+ were estimated for the 2010 FW and NMRW samples. The average NFs were in the order of Ca2+ (2.8) >, K+ (0.6) > Mg2+ (0.4) > NH4+ (0.3) in fog water. For the non-monsoonal rainwater, the NFs order were Ca2+ (1.8)>Mg2+ (0.6)>NH4+ (0.4)>K+ (0.1). Ca2+ (70 and 57%) was the dominant neutralizing ion in both FW and NMRW, respectively. The second highest NF in FW was K+ (9%). However, in RW, Mg2+ was the next highest (27%). The present study indicates that there is little possibility of acidic fogs or rainwater in northern India given the high concentrations of alkaline species in the atmosphere.
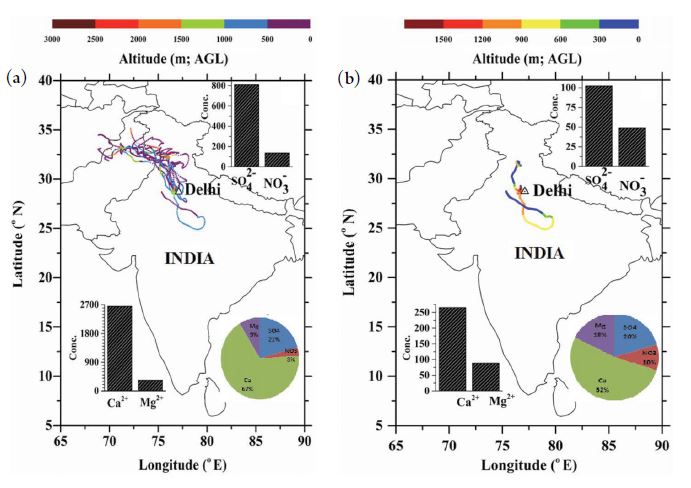
3. 5 Air Mass Back Trajectories
Five-day air-mass back trajectories ending over the receptor site in New Delhi were employed to examine the transportation of atmospheric pollutants over the study region. The Hybrid Single-Particle Lagrangian Integrated Trajectory (HYSPLIT 4 Model) model was used to assess the data via NOAA ARL READY Website (https://www.arl.noaa.gov/HYSPLIT) and NOAA Air Resources Laboratory, Silver Spring, MD with Global Data Assimilation System data (Rolph et al., 2017; Stein et al., 2015). The vertical motion model was chosen. The trajectories for FW (Fig. 5a) and NMRW (Fig. 5b) help to identify the likely source regions for the chemical constituents. Wang et al. (2011) reported that the concentrations of dissolved ions (both anions and cations) in cloud water vary greatly depending on the source region. In the present study, it was observed that there were two different types of air masses, i.e., air masses originating from the northwest direction (92%) and for a few cases (8%), air masses originating from the southeast direction. To the northwest, the air masses originated from Afghanistan, Pakistan, touching the highly fertile region of Punjab and Haryana states before reaching the sampling site. During the non-monsoon periods, very little rainfall (only two days) was recorded. These events were due to western disturbances with air masses from the north of the Indian subcontinent on one day and from the other direction (southeast) on the other day. The concentrations of Cl-, K+, Ca2+, and Mg2+ in the FW were 92, 45, 73, and 82% higher when the air masses arrive from the southeast on 24th January 2016. NH4+ was reduced by about 32%. The FW acidic species such as SO42- and NO3- were less variability with direction. In RW, the concentrations of Ca2+ and SO42- were a factor of two and three times higher, respectively, when the air masses arrived from the north. Potassium was much higher when the air mass arrived from the northwest direction. In RW, ammonium and nitrate were around double and 48% higher, respectively, when the air masses were from the southeast. The concentrations of Ca2+ and SO42- were 10 and 8 times higher, respectively, in the FW compared to NMRW. However, the ratios of Ca2+ to SO42- in FW and NMRW were about three in both rain and fog water. The contribution of acidic species (SO42- and NO3-) in FW and NMRW were ~24% and 30%, respectively. Soil species (Ca2+ and Mg2+) were around 76 and 60%, respectively (Fig. 3a, b).
4. CONCLUSIONS
Concentrations of ionic species in fog water (FW) and non-monsoonal rainwater (NMRW) have changed somewhat over the period from 1985 to 2010. This paper summarizes the ionic composition of FW and NMRW collected in Delhi in northern India. Changes in population and emission sources have led to many-fold increases in concentrations of secondary species such as sulfate. During the study period, the TMIC in FW and NMRW were 5,738 and 814 μeq L-1, respectively, indicating highly concentrated FW in northern India. The TMIC in FW is approximately a factor of three (2,040 μeq L-1) and 88% (3,050 μeq L-1) higher than the 2001/2002 and 1985 values. The concentrations of acidic species (SO42- and NO3-) in FW were much higher than that of MRW (3 and 5 times) and NMRW (8 and 12 times), respectively. The concentrations of SO42- and NO3- were around double in NMRW compared to MRW indicating the higher loading of acidic species during the winter period. Significant decadal growth in the mean concentrations of species in FW (SO42- - about 9 times; NH4+ - double) was observed from 1985 to 2010. Nitrate concentrations were reduced by about 28%. The higher sulfate contribution in FW and NMRW is likely due to the continued use of heavy-duty vehicles burning high sulfur fuel. The contribution of anions in FW, MRW, and NMRW were 20, 42, and 43% however, the cation contributions were 80, 58, and 57%, respectively. The contribution of anions was much lower in FW compared to MRW and NMRW indicating weaker scavenging of anions by fog. The alkaline nature of the soils in this region suggests that acid precipitation is unlikely to be observed in this region. This work suggests that improved fuel quality will be required to substantially reduce the local sulfate concentrations.
Acknowledgments
The Ministry of Earth Sciences (MoES), Government of India, New Delhi, supports the Indian Institute of Tropical Meteorology (IITM), Pune. The authors gratefully acknowledge the NOAA Air Resources Laboratory (ARL) for the provision of the HYSPLIT transport and dispersion model (http://www.ready.noaa.gov) used in this publication.
References
-
Ali, K., Momin, G.A., Tiwari, S., Safai, P.D., Chate, D.M., Rao, P.S.P. (2004) Fog and Precipitation Chemistry at Delhi, North India. Atmospheric Environment, 38, 4215-4222.
[https://doi.org/10.1016/j.atmosenv.2004.02.055]

-
Badarinath, K.V.S., Kharol, S.K., Sharma, A.R., Roy, P.S. (2009) Fog over Indo-Gangetic plains-a study using multi-satellite data and ground observations. IEEE Journal of Selected Topics in Applied Earth Observations and Remote Sensing, 2(3), 185-195.
[https://doi.org/10.1109/JSTARS.2009.2019830]

-
Bisht, D.S., Tiwari, S., Srivastava, A.K., Singh, J.V., Singh, B.P., Srivastava, M.K. (2015) High concentration of acidic species in rainwater at Varanasi in the Indo-Gangetic Plains, India. Natural Hazards, 75, 2985-3003.
[https://doi.org/10.1007/s11069-014-1473-0]

-
Budhavant, K.B., Rao, P.S.P., Safai, P.D., Granat, L., Rodhe, H. (2014) Chemical composition of the inorganic fraction of cloud-water at a high-altitude station in West India. Atmospheric Environment, 88, 59-65.
[https://doi.org/10.1016/j.atmosenv.2014.01.039]

-
Butler, C.D., Trumble, J.T. (2008) Effects of pollutants on bottom-up and top-down processes in insect-plant interactions. Environment Pollution, 156, 1-10.
[https://doi.org/10.1016/j.envpol.2007.12.026]

- Census of India (2011) Available at: http://censusindia.gov.in/3622011-prov-results/provdata_productsdelhi.html.
-
Charlson, R., Rodhe, H. (1982) Factors controlling the acidity of natural rainwater. Nature, 295, 683-685.
[https://doi.org/10.1038/295683a0]

-
Chate, D.M., Ghude, S.D., Beig, G., Mahajan, A., Jena, S., Srinivas, C.K., Dahiya, R., Kumar, A.N. (2014) Deviations from the O3-NO-NO2 photo-stationary state in Delhi, India. Atmospheric Environment, 96, 353-358.
[https://doi.org/10.1016/j.atmosenv.2014.07.054]

-
Chelani, A.B. (2012) Persistence Analysis of Extreme CO, NO2 and O3 Concentrations in Ambient Air of Delhi. Atmospheric Research, 108, 128-134.
[https://doi.org/10.1016/j.atmosres.2012.02.001]

-
Collett Jr., J.L., Herckes, P., Youngster, S., Lee, T. (2008) Processing of atmospheric organic matter by California radiation fogs. Atmospheric Research, 87, 232-241.
[https://doi.org/10.1016/j.atmosres.2007.11.005]

-
Collett Jr., J.L., Sherman, D.E., Moore, K.F., Hannigan, M.P., and Lee, T. (2001) Aerosol particle processing and removal by fogs: observations in chemically heterogeneous central California radiation fogs. Water, Air and Soil Pollution: Focus, 1, 303-312.
[https://doi.org/10.1023/A:1013175709931]

-
Cong, Z., Kawamura, K., Kang, S., Fu, P. (2015) Penetration of biomass-burning emissions from South Asia through the Himalayas: new insights from atmospheric organic acids. Scientific Reports, 5, 9580.
[https://doi.org/10.1038/srep09580]

-
Das, N., Das, R., Chaudhury, G.R., Das, S.N. (2010) Chemical composition of precipitation at background level. Atmospheric Research, 95, 108-113.
[https://doi.org/10.1016/j.atmosres.2009.08.006]

-
Deshmukh, D.K., Kawamura, K., Gupta, T., Haque, M.M., Zhang, Y.L., Singh, D.K., Tsai, Y.I. (2019) High Loadings of Water-soluble Oxalic Acid and Related Compounds in PM2.5 Aerosols in Eastern Central India: Influence of Biomass Burning and Photochemical Processing. Aerosol Air Quality Research, 19, 2625-2644.
[https://doi.org/10.4209/aaqr.2019.10.0543]

-
Falkovich, A.H., Graber, E.R., Schkolnik, G., Rudich, Y., Maenhaut, W., Artaxo, P. (2005) Low molecular weight organic acids in aerosol particles from Rondônia, Brazil, during the biomass-burning, transition and wet periods. Atmospheric Chemistry and Physics, 5, 781-797.
[https://doi.org/10.5194/acp-5-781-2005]

-
Fang, Z., Li, C., He, Q., Czech, H., Gröger, T., Zeng, J., Fang, H., Xiao, S., Pardo, M., Hartner, E., Meidan, D., Wang, X., Zimmermann, R., Laskin, A., Rudich, Y. (2021) Secondary organic aerosols produced from photochemical oxidation of secondarily evaporated biomass burning organic gases: Chemical composition, toxicity, optical properties, and climate effect. Environment International, 157, 106801.
[https://doi.org/10.1016/j.envint.2021.106801]

- Fukuzaki, N., Khodzher, T.V., Haras, H. (2002) Quality Control of Chemical Analysis in Acid Deposition Monitoring. Improvement in Ion Balance by Hydrogenecarbonate Determination. Journal of Japan Society for Atmospheric Environment. 37(6), 393-401.
-
Gilardoni, S., Massoli, P., Giulianelli, L., Rinaldi, M., Paglione, M., Pollini, F., Lanconelli, C., Poluzzi, V., Carbone, S., Hillamo, R., Russell, L.M., Facchini, M.C., Fuzzi, S. (2014) Fog scavenging of organic and inorganic aerosol in the Po Valley. Atmospheric Chemistry and Physics, 14, 6967-6981.
[https://doi.org/10.5194/acp-14-6967-2014]

-
Gillett, R.W., Ayers, G.P. (1991) The use of thymol as a biocide in rainwater samples. Atmospheric Environment. Part A. General Topics, 25(12), 2677-2681.
[https://doi.org/10.1016/0960-1686(91)90198-G]

-
Gultepe, I., Tardif, R., Michaelides, S.C., Cermak, J., Bott, A., Bendix, J., Müller, M.D., Pagowski, M., Hansen, B., Ellrod, G., Jacobs, W., Toth, G., Cober, S.G. (2007) Fog research: a review of past achievements and future perspectives. Pure and Applied Geophysics, 164, 1121-1159.
[https://doi.org/10.1007/s00024-007-0211-x]

-
Herckes, P., Marcotte, A., Wang, Y., Collett, J.L. (2015) Fog Composition in the Central Valley of California over Three Decades. Atmospheric Research, 151, 20-30.
[https://doi.org/10.1016/j.atmosres.2014.01.025]

-
Igawa, M., Matsumura, K., Okochi, H. (2001) Fog water chemistry at Mt. Oyama and its dominant factors. Water, Air and Soil Pollution, 130, 607-612.
[https://doi.org/10.1023/A:1013867620159]

- IPCC 2014: Climate change (2014) Synthesis Report. Contribution of working groups I, II, and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change, Switzerland, 151p.
-
Jacob, D.J., Shair, F.H., Waldman, J.M., Munger, W.M., Hoffmann, M.R. (1987) Transport and oxidation of SO2 in a stagnant foggy valley. Atmospheric Environment, 21, 1305-1314.
[https://doi.org/10.1016/0004-6981(67)90077-7]

-
Jacobson, J.S. (1984) Effects of acidic aerosol, fog, mist and rain on crops and trees. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, 305, 327-338.
[https://doi.org/10.1098/rstb.1984.0061]

-
Jain, M., Kulshrestha, U., Sarkar, A.K., Parashar, D.C. (2000) Influence of crustal aerosols on wet deposition at urban and rural sites in India. Atmospheric Environment, 34, 5129-5137.
[https://doi.org/10.1016/S1352-2310(00)00350-2]

- Jenamani, R.K., Rathore, L.S. (2015) Physical processes from data analysis, realtime forecast system, performances of various indigenous fog models and bench marking, FDP-FOG 2008-2015, Fog Tech Report 2/2015.
-
Kapoor, R.K., Tiwari, S., Ali, K., Singh, G. (1993) Chemical analysis of fog water at Delhi, North India. Atmospheric Environment, 27A, 2453-2455.
[https://doi.org/10.1016/0960-1686(93)90415-U]

-
Kaskaoutis, D.G., Kumar, S., Sharma, D., Singh, R.P., Kharol, S.K., Sharma, M., Singh, A.K., Singh, S., Singh, A., Singh, D. (2014) Effects of crop residue burning on aerosol properties, plume characteristics and long range transport over northern India. Journal of Geophysical Research, 119, 5424-5444.
[https://doi.org/10.1002/2013JD021357]

-
Khemani, L.T., Momin, G.A., Rao, P.S.P., Safai, P.D., Prakash, P. (1987) Influence of alkaline particulates on the chemistry of fog water at Delhi, North India. Water, Air, and Soil Pollution, 34, 183-189.
[https://doi.org/10.1007/BF00184759]

-
Khemani, L.T., Momin, G.A., Rao, P.S.P., Safai, P.D., Singh, G., Kapoor, R.K. (1989) Spread of acid rain over India. Atmospheric Environment (1967), 23(4), 757-762.
[https://doi.org/10.1016/0004-6981(89)90478-2]

-
Kulshrestha, U.C., Kulshrestha, M.J., Sekar, R., Sastry, G.S.R., Vairamani, M. (2003) Chemical characteristics of rainwater at an urban site of south-central India. Atmospheric Environment, 37, 3019-3026.
[https://doi.org/10.1016/S1352-2310(03)00266-8]

-
Lange, C.A., Matschullat, J., Zimmermann, F., Stertig, G., Wienhaus, O. (2003) Fog chemistry and chemical composition of fog water - a relevant contribution to atmospheric deposition in the eastern Erzgebirge, Germany. Atmospheric Environment, 37, 3731-3739.
[https://doi.org/10.1016/S1352-2310(03)00350-9]

-
Langner, J., Rodhe, H. (1991) A global 3-dimensional model of the tropospheric sulfur cycle. Journal of Atmospheric Chemistry, 13, 225-263.
[https://doi.org/10.1007/BF00058134]

-
Li, P., Li, X., Yang, C., Wang, X., Chen, J., Collett Jr., J.L. (2011) Fog Water Chemistry in Shanghai. Atmospheric Environment, 45, 4034-4041.
[https://doi.org/10.1016/j.atmosenv.2011.04.036]

-
Lu, C., Niu, S., Tang, L., Lv, J., Zhao, L., Zhu, B. (2010) Chemical Composition of Fog Water in Nanjing area of China and its Related Fog Microphysics. Atmospheric Research, 97, 47-69.
[https://doi.org/10.1016/j.atmosres.2010.03.007]

-
Munger, J.W., Jacob, D.J., Waldman, J.M., Hoffmann, M.R. (1983) Fog water chemistry in an urban atmosphere. Journal of Geophysical Research, 88, 5109-5121.
[https://doi.org/10.1029/JC088iC09p05109]

-
Mohan, K.N., Paligan, A.A., Sivakumar, G., Krishnamurthy, R., Shubha, V., Shinde, U., Mali, R.R., Bhatnagar, M.K. (2015) Performance study of Drishti transmissometer at CAT III B airport. Mausam, 66(4), 713-718.
[https://doi.org/10.54302/mausam.v66i4.578]

-
Nagpure, A.S., Sharma, K., Gurjar, B.R. (2013) Traffic induced emission estimates and trends (2000-2005) in megacity Delhi. Urban Climate, 4, 61-73.
[https://doi.org/10.1016/j.uclim.2013.04.005]

-
Niu, F., Li, Z., Li, C., Lee, K.H., Wang, M. (2010) Increase of wintertime fog in China: potential impacts of weakening of the Eastern Asian monsoon circulation and increasing aerosol loading. Journal of Geophysical Research, 115, D00K20.
[https://doi.org/10.1029/2009JD013484]

-
Pachon, J.E., Weber, R.J., Zhang, X., Mulholland, J.A., Russell, A.G. (2013) Revising the use of potassium (K) in the source apportionment of PM2.5. Atmospheric Pollution Research, 4, 14-21.
[https://doi.org/10.5094/APR.2013.002]

-
Pandis, S.N., Seinfeld, J.H., Pilinis, C. (1990) The smog-fogsmog cycle and acid deposition. Journal of Geophysical Research, 95, 18489-18500.
[https://doi.org/10.1029/JD095iD11p18489]

-
Perrino, C., Catrambone, M., Dalla Torre, S., Rantica, E., Sargolini, T., Canepari, S. (2014) Seasonal variations in the chemical composition of particulate matter: a case study in the Po Valley. Part I: macro-components and mass closure. Environmental Science and Pollution Research, 21, 3999-4009.
[https://doi.org/10.1007/s11356-013-2067-1]

-
Pruppacher, H.R., Klett, J.D. (2010) Microphysics of Clouds and Precipitation, Second ed. Springer, Dordrecht, the Netherlands.
[https://doi.org/10.1007/978-0-306-48100-0]

-
Raja, S., Raghunathan, R., Kommalapati, R.R., Shen, X., Collett, J.L., Valsaraj, K.T. (2009) Organic composition of fogwater in the Texas-Louisiana gulf coast corridor. Atmospheric Environment, 43, 4214-4222.
[https://doi.org/10.1016/j.atmosenv.2009.05.029]

-
Rolph, G., Stein, A., Stunder, B. (2017) Real-time Environmental Applications and Display sYstem: READY. Environmental Modelling & Software, 95, 210-228.
[https://doi.org/10.1016/j.envsoft.2017.06.025]

-
Sahu, S.K., Beig, G., Parkhi, N.S. (2011) Emissions inventory of anthropogenic PM2.5 and PM10 in Delhi during Commonwealth Games 2010. Atmospheric Environment, 45, 6180-6190.
[https://doi.org/10.1016/j.atmosenv.2011.08.014]

-
Sahu, S.K., Beig, G., Parkhi, N.S. (2015) High Resolution Emission Inventory of NOx and CO for Mega City Delhi, India. Aerosol Air Quality Research, 15, 1137-1144.
[https://doi.org/10.4209/aaqr.2014.07.0132]

-
Sander, S., Seinfeld, J. (1976) Chemical Kinetics of Homogeneous Atmospheric Oxidation of SO2. Environmental Science & Technology, 10, 1114-1123.
[https://doi.org/10.1021/es60122a007]

- Seinfeld, J.H., Pandis, S.N. (2006) Atmospheric Chemistry and Physics - From Air Pollution to Climate Change, 2nd edition, Wiley Inter-science.
- Senaratne, I., Kelliher, F., Triggs, C. (2005) Source Apportionment of PM10 During Cold, Calm Weather in Christchurch, New Zealand: Preliminary Results from a Receptor Model. Clean Air and Environmental Quality, 39(3), 47-54. https://search.informit.org/doi/10.3316/informit.373033066203184
-
Shigihara, A., Matsumura, Y., Kashiwagi, M., Matsumoto, K., Igawa, M. (2009) Effects of acidic fog and ozone on the growth and physiological functions of Fagus crenata saplings. Journal of Forest Research, 14, 394-399.
[https://doi.org/10.1007/s10310-009-0144-6]

-
Sickles II, J.E., Hodson, L.L., Vorburger, L.M. (1999) Evaluation of the filter pack for long-duration sampling of ambient air. Atmospheric Environment, 33, 2187-2202.
[https://doi.org/10.1016/S1352-2310(98)00425-7]

-
Simon, S., Klemm, O., El-Madany, T., Walk, J., Amelung, K., Lin, P.H., Chang, S.C., Lin, N.H., Engling, G., Hsu, S.C., Wey, T.H., Wang, Y.N., Lee, Y.C. (2016) Chemical Composition of Fog Water at Four Sites in Taiwan. Aerosol Air Quality Research, 16, 618-631.
[https://doi.org/10.4209/aaqr.2015.03.0154]

- Srinivas, B., Kumar, R., Surya Prakash Rao, K. (2001) A study on the chemistry of rainwater in and around Hyderabad and Secunderabad. Indian Journal of Environmental Protection, 21, 210-213.
-
Srivastava, A., Jain, V.K. (2008) Source apportionment of suspended particulate matters in a clean area of Delhi: A note. Transportation Research Part D: Transport and Environment, 13, 59-63.
[https://doi.org/10.1016/j.trd.2007.09.001]

-
Stein, A.F., Draxler, R.R., Rolph, G.D., Stunder, B.J.B., Cohen, M.D., Ngan, F. (2015) Noaa’s hysplit atmospheric transport and dispersion modeling system. American Meteorological Society. 2015, 96, 2059-2077.
[https://doi.org/10.1175/BAMS-D-14-00110.1]

-
Sun, X., Wang, Y., Li, H., Yang, X., Sun, L., Wang, X., Wang, T., Wang, W. (2016) Organic acids in cloud water and rainwater at a mountain site in acid rain areas of South China. Environmental Science and Pollution Research, 23, 9529-9539.
[https://doi.org/10.1007/s11356-016-6038-1]

-
Tiwari, S., Payra, S., Mohan, M. (2011) Visibility Degradation during Foggy Period due to Anthropogenic Urban Aerosol at Delhi, India. Atmospheric Pollution Research, 2, 116-120.
[https://doi.org/10.5094/APR.2011.014]

-
Tiwari, S., Chate, D.M., Bisht, D.S., Srivastava, M.K., Padmanabhamurty, B. (2012) Rainwater chemistry in the North Western Himalayan region of India. Atmospheric Research, 104, 128-138.
[https://doi.org/10.1016/j.atmosres.2011.09.006]

-
Tiwari, S., Pandithurai, G., Attri, S.D., Srivastava, A.K., Soni, V.K., Bisht, D.S., Kumar, V.A., Srivastava, M.K. (2015) Aerosol optical properties and their relationship with meteorological parameters during wintertime in Delhi, India. Atmospheric Research, 153, 465-479.
[https://doi.org/10.1016/j.atmosres.2014.10.003]

-
Tiwari, S., Hopke, P.K., Thimmaiah, D., Dumka, U.C., Srivastava, A.K., Bisht, D.S., Rao, P.S.P., Chate, D.M., Srivastava, M.K., Tripathi, S.N. (2016a) Nature and Sources of Ionic Species in Precipitation across the Indo-Gangetic Plains, India. Aerosol Air Quality Research, 16, 943-957.
[https://doi.org/10.4209/aaqr.2015.06.0423]

-
Tiwari, S., Tiwari, S., Hopke, P.K., Attri, S.D., Soni, V.K., Singh, A.K. (2016b) Variability in optical properties of atmospheric aerosols and their frequency distribution over a mega city “New Delhi”, India. Environmental Science and Pollution Research, 23, 8781-8793.
[https://doi.org/10.1007/s11356-016-6060-3]

-
Waldman, J.M., Munger, J.W., Jacob, D.J., Flagan, R.C., Morgan, J.J., Hoffmann, M.R. (1982) Chemical composition of acid fog. Science, 218, 677-680.
[https://doi.org/10.1126/science.218.4573.677]

-
Walter, J.D. (1983) The Quantity of Acid in Acid Fog. Journal of the Air Pollution Control Association, 33, 691-692.
[https://doi.org/10.1080/00022470.1983.10465629]

-
Wang, B., Li, T. (1994) Convective interaction with boundary-layer dynamics in the development of a tropical intraseasonal system. Journal of the Atmospheric Sciences, 51, 1386-1400.
[https://doi.org/10.1175/1520-0469(1994)051<1386:CIWBLD>2.0.CO;2]

-
Wang, Y., Guo, J., Wang, T., Ding, A., Gao, J., Zhou, Y., Collett Jr., J., Wang, W. (2011) Influence of regional pollution and sandstorms on the chemical composition of cloud/fog at the summit of Mt. Taishan in northern China. Atmospheric Research, 99, 434-442.
[https://doi.org/10.1016/j.atmosres.2010.11.010]

-
Wang, Y., Zhang, J., Marcotte, A.R., Karl, M., Dye, C., Herckes, P. (2015) Fog Chemistry at Three Sites in Norway. Atmospheric Research, 151, 72-81.
[https://doi.org/10.1016/j.atmosres.2014.04.016]

-
Watanabe, K., Honoki, H., Iwama, S., Iwatake, K., Mori, S., Nishimoto, D., Uehara, Y. (2011) Chemical composition of fog water at Mt. Tateyama near the coast of the Japan Sea. Erdkunde, 65, 233-245. https://www.jstor.org/stable/23069696
[https://doi.org/10.3112/erdkunde.2011.03.02]

-
Watanabe, K., Ishizuka, Y., Minami, Y., Yoshida, K. (2001) Peroxide concentrations in fog water at mountainous sites in Japan. Water, Air Soil Pollution, 130, 1559-1564.
[https://doi.org/10.1023/A:1013974728609]

-
Watanabe, K., Takebe, Y., Sode, N., Igarashi, Y., Takahashi, H., Dokiya, Y. (2006) Fog and Rain Water Chemistry at Mt. Fuji: A Case Study during the September 2002 Campaign. Atmospheric Research, 82, 652-662.
[https://doi.org/10.1016/j.atmosres.2006.02.021]

-
Weatherburn, M.W. (1967) Phenol-hypochlorite reaction for determination of ammonia. Analytical Chemistry, 39, 971-974.
[https://doi.org/10.1021/ac60252a045]